
It does not complete the octet rule like other elements. Boron (B) has three single electrons, so it can form three covalent bonds. This can be seen by drawing lewis dot structures of elements.įor example, hydrogen (H) has a single electron so it can form a covalent bond by completing the doublet rule. It is because atoms form covalent bonds to complete their doublet or octet rule. By counting the number of single dots or electrons on the elements, we can predict the diagram of the element. Lewis dot structures have single and double dots representing unpaired and paired electrons. This trend is followed by all groups in the periodic table, groups 5A, 6A, 7A, and 8A. Similarly, carbon (C) and silicon (Si) have four valence electrons and belong to group 4A. For example, boron (B) has three valence electrons in its outermost shell and belongs to group 3A of the periodic table. These valence electrons are equal to the group of the element in the periodic table. They have fixed numbers of electrons in their outermost shell called valence electrons. However, non-metals have filled s-orbitals and partially filled p-orbitals. Hydrogen is an exceptional case and when combined with non-metallic elements forms a covalent compound. Metallic elements can lose electrons and form ions. That’s why it is better to use one type of method, lewis structure or lewis dot structure to demonstrate the paired and unpaired electrons of molecules or compounds. These electrons are not fixed on a specific side of an atom, they can be drawn on the random sides of elements.īoth lewis structure and lewis dot structure are valid to represent the structure of molecules but it creates confusion when we use the solid lines for unpaired shared electrons and dots for paired electrons. The whole structure is a combination of dots that represents electrons. However, in the lewis dot structures, paired and unpaired electrons are shown in the form of dots. When a structure of a molecule is drawn and paired electrons are shown in the form of dots and unpaired shared electrons are shown in the form of a solid line called a lewis structure, not a lewis dot structure. The only difference is the mode of drawing or presenting the structures of molecules or compounds. The concept is the same for both terminologies which are lewis structure and lewis dot structure. They don’t take part in the formation of bonds between the atoms of a molecule or compound. However, double dots show as a pair of electrons on atoms in a molecule called lone pair. Now, these unpaired shared electrons are called bond pairs. This electron is responsible for the chemical bond formation when it comes close to the unpaired electron of other atoms. A single dot represents an unpaired electron. These diagrams are named lewis dot structures because valence electrons are drawn in the form of dots around the four sides of atoms. They have valence electrons that are manually arranged around the sides of the symbol of elements. 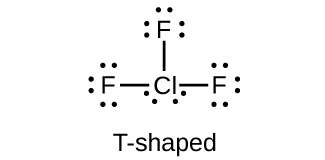
Lewis dot structures are the chemical symbols of elements. As a result of his research, he discovered covalent bonds and developed a method to represent the bonds between the atoms of molecules by using simple structures called lewis electrons or lewis dot structures. Please comment below or contact us.In 1916, an American chemist Gilbert Newton Lewis worked on how atoms are connected in a molecule. The ions are atoms that have gained one or more electrons (known as anions, which are negatively charged) and atoms that have lost one or more electrons (known as cations, which are positively charged).Ī covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding. Ionic bonding is a type of chemical bond that involves the electrostatic attraction between oppositely charged ions, and is the primary interaction occurring in ionic compounds. What is chemical bond, ionic bond, covalent bond?Ī chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds. Question: Is Ba(ClO4)2 an ionic or covalent bond ?Īnswer: Ba(ClO4)2 ( Barium perchlorate ) is an ionic bond
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
